Cancer is personal.
Cancer therapy should be, too.
Precision oncology powered by AI

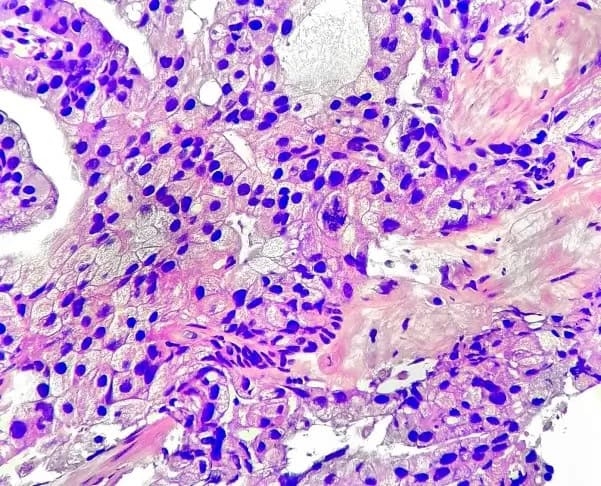
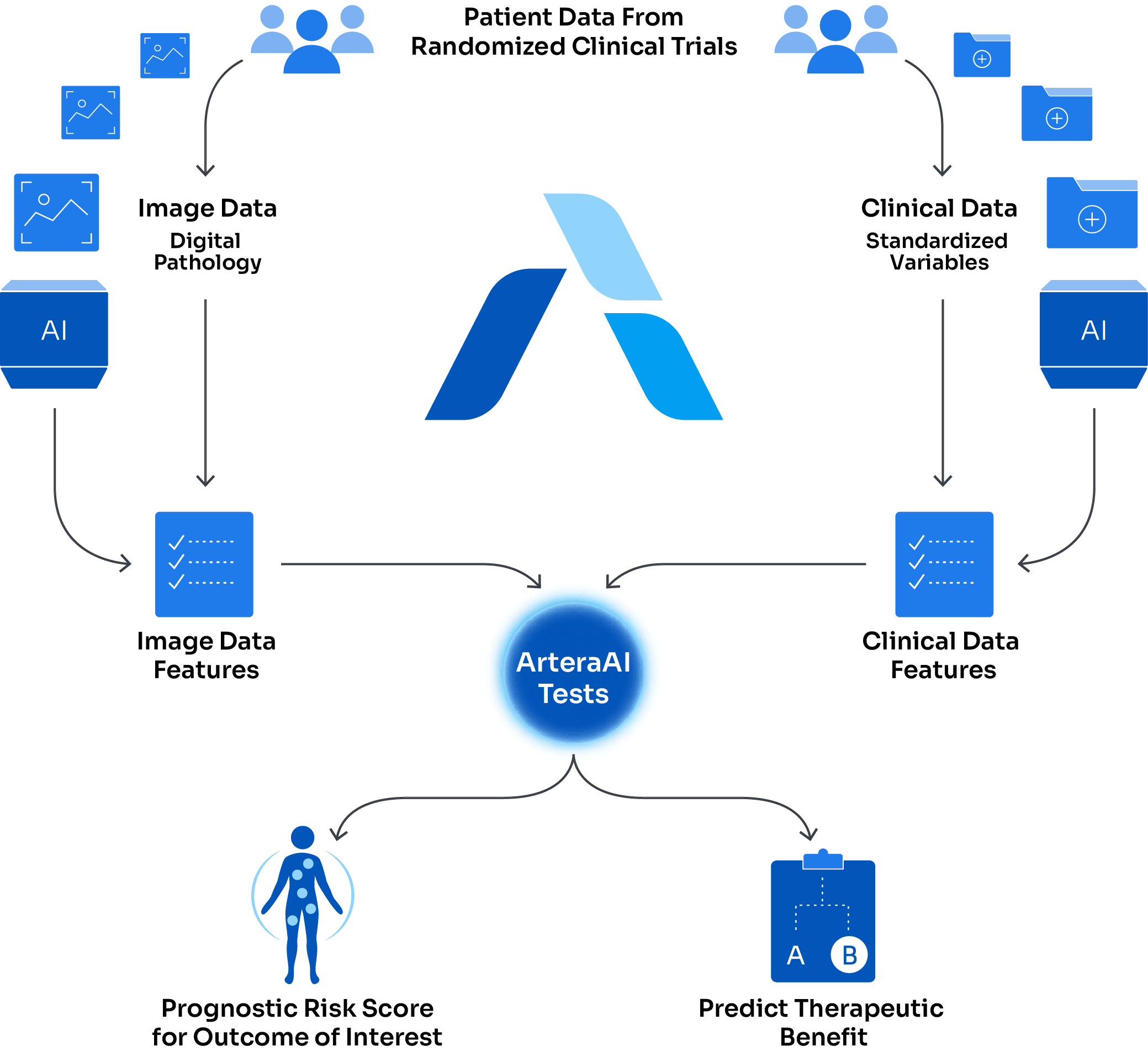
Using AI to personalize treatment
- Artera is a leader in AI-powered precision medicine
- Multi-modal artificial intelligence (MMAI) platform analyzes digital pathology and clinical data for personalized cancer management
- Validated in dozens of large, randomized phase III clinical trials
FDA Cleared
Following an earlier Breakthrough Device Designation, Artera achieved De Novo Authorization from the FDA for ArteraAI Prostate, a first-of-its-kind AI-powered digital pathology risk stratification tool now regulated as Software as a Medical Device (SaMD).
Actionable insights across multiple cancers
Prostate Cancer
ArteraAI Prostate Test offers therapeutic insights in early-stage prostate cancer and can predict which NCCN Intermediate-risk patients will benefit from ST-ADT with their radiation therapy. It is the first and only AI test to be recommended in the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for Prostate Cancer.
-

Does not consume tissue
Analysis uses image data from existing H&E slides with no additional staining or preparation required
-

Rapid turnaround
Results can be shared within 1-2 days after receipt of the patient’s specimen by our CLIA lab
-

Represents diverse populations
Consistent prognostic performance between African American and non-African American men -
 Learn More
Learn MoreCovered by insurance
Test is fully reimbursed for Medicare patients and many commercial health plans provide coverage for the test
Breast Cancer
The ArteraAI Breast Cancer Test provides 5- and 10-year distant metastasis risk for early-stage breast cancer patients and predicts which node-negative patients 50 years or older will benefit from chemotherapy.
We’re passionate about personalizing cancer care
Driven by integrity, innovation, and the advancement of medical technology, our team of experts is passionate about developing AI tools that help enable clinicians to deliver individualized treatment plans.
Order the ArteraAI
Prostate Test for
your patient.
Contact Us














