We are offering select clinicians the opportunity to participate in an Early Access Program in exchange for their feedback on the test report. If you are interested in being part of this program, you can add your name to the waitlist here.
The Science Behind the ArteraAI Breast Cancer Test
Artera’s multimodal artificial intelligence (MMAI) platform leverages a unique algorithm that assesses both the digital images from a patient’s resection and their clinical data. The AI combines this information to estimate long-term outcomes and predict whether a patient will benefit from additional therapy.
The algorithm was developed using large datasets, from thousands of patients and tens of thousands of pathology slide images, and has been clinically validated using multiple Phase 3 randomized trials and in a variety of patient cohorts.
Ordering is Easy with Artera
The ArteraAI Breast Cancer Test is a laboratory-developed testTesting is performed by Artera, located at 6800 Southpoint Pkwy Suite 950, Jacksonville, Florida 32216. The ArteraAI Breast Cancer Test was developed and its performance characteristics were determined by Artera. This Laboratory is certified under the Clinical Laboratory Improvement Amendments of 1988 (CLIA ‘88) as qualified to perform high-complexity clinical laboratory testing. This test is used for clinical purposes and should not be regarded as investigational or for research. This test has not been cleared or approved by the U.S. Food and Drug Administration.. that is now clinically available through a single CLIA-certified laboratory in Jacksonville, FL.
The ArteraAI Breast Cancer Test is intended to assist clinicians with risk-based decisions in female patients with HR+/HER2- early stage invasive breast cancer within recommended clinical guidelines. The test is intended to be ordered after tumor resection for patients who have not received neoadjuvant therapy prior to surgery.
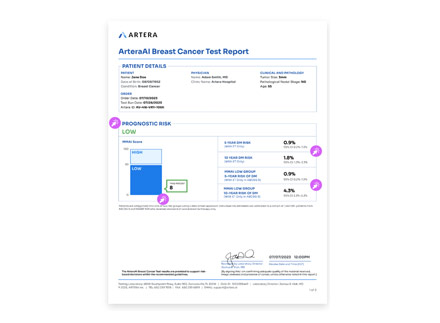
Yes! You can visit the ArteraAI Breast Cancer Test report page to explore each section of the report to better understand the results.
The ArteraAI Breast Cancer Test can be ordered in 49 states, the lone exception being New York. We also have a partner the United Kingdom where a version of the ArteraAI Breast Cancer Test is available. Please reach out to support@artera.ai for additional details.
No. Ordering of the ArteraAI Breast Cancer Test is limited to clinicians whose license and scope of practice allows them to order the test. However, we encourage patients to share information about the ArteraAI Breast Cancer Test with their doctor.
Using the ArteraAI Breast Cancer Test, results are typically ready within 1-2 days after receipt of the patient’s specimen. The patient’s report will be faxed to the ordering clinician.
The out-of-pocket cost for the ArteraAI Breast Cancer Test will be based on the patient’s insurance plan. Patients should contact billing@artera.ai for additional questions about their estimated out-of-pocket liability for the test.
We offer a Financial Assistance Program to help reduce the potential out of pocket costs associated with testing. Please contact billing@artera.ai to learn more.
For Early Access Program participants, tests can be ordered with zero out of pocket costs to patients. To learn more about this program, please fill out this form.
Order the ArteraAI Breast Cancer Test for your patient. Start your order